Chemical Stability of Coconut Shell Biochar Under Oxidative Conditions
| Received 05 Oct, 2025 |
Accepted 20 Jan, 2026 |
Published 21 Jan, 2026 |
Background and Objective: Biochar, a carbon-rich byproduct of biomass pyrolysis, holds promise for soil enhancement, water purification, and environmental remediation, largely due to its carbon sequestration potential. However, the stability of carbon within biochar varies, influencing its effectiveness. This study aimed to evaluate the relationship between pyrolysis temperature and the chemical oxidative stability of coconut shell-derived biochar. Materials and Methods: Coconut shell biomass was pyrolyzed at four different temperatures (400, 500, 600, and 700°C). The resulting biochar samples were subjected to chemical oxidation using potassium permanganate (KMnO4) at neutral pH (7.2) to assess stability. Stability was quantified as a percentage and compared across treatments. Ultimate analysis and atomic ratios (H:C and O:C) were used to support interpretations of aromaticity and hydrophobicity. Results: Biochar stability increased with rising pyrolysis temperature, indicating enhanced oxidative resistance at higher heat treatments. These findings aligned with elemental data and decreasing H:C and O:C ratios, suggesting increased aromatic carbon content and hydrophobicity. Conclusion: Higher pyrolysis temperatures improved the oxidative stability of coconut shell biochar due to increased aromatic structure and reduced oxygen-containing groups. These findings support the use of high-temperature biochar in long-term environmental applications, although further field-based validation is recommended.
| Copyright © 2026 Ejimadu et al. This is an open-access article distributed under the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited. |
INTRODUCTION
Biochar, the C-rich solid product of pyrolysis of biomass, is usually characterized by its porous structures, large surface area, high thermal stability, and multiple surface functional groups. On account of its potential as a multifunctional material that could be useful in carbon sequestration, soil quality improvement, water treatment, and environmental remediation1,2, biochar has become of increasing multidisciplinary research3. However, some carbon atoms in biochar are quite unstable and are relatively easily released into the environment in various ways4, with consequences on its overall stability and effectiveness in carbon sequestration. Hence, assessing the chemical oxidative stability of biochar is imperative. Oxidation stability is of particular relevance as it influences the long-term stability of biochar in soil. The methods frequently used to estimate the long term stability of biochar in the environment rely on chemical analysis; proximate; volatile matter, ash content, moisture content and fixed carbon, and ultimate; C, H, O, N and S contents of the biochar, followed by estimation of various elemental ratios (indices) H:C, O:C, etc. that indicate aromaticity and polarity in the biochar and hence its stability and recalcitrance to degradation5,6.
Therefore, methods that are more indicative of stability are required to determine the carbon sequestration potential. Direct chemical oxidation methods involving extraction with several chemicals, hot water, acids, and alkalis, and accelerated oxidation methods using hydrogen peroxide and potassium dichromate, have been proposed7,8. The coconut industry generates more than 124 million tons of waste, coconut husks, and coconut shells annually9. The waste materials are often disposed of indiscriminately into the environment and/or burnt in open air, causing both public health and environmental concerns10. Pyrolysis, a thermochemical process, offers a unique method for obtaining useful end-products: Biogas, bio-oil, and biochar from biomass. Pyrolysis conditions, particularly, highest heat treatment temperature (HTT), are an important factor that influences biochar properties and applications.
In this work, different pyrolysis temperatures were used to produce coconut shell biochar, and the resistance (recalcitrance) to chemical oxidation through an accelerated oxidation technique, followed by elemental analysis of the oxidised biochar, was determined. There are no published reports that specifically explored the effect of pyrolysis temperature as a function of the chemical oxidation stability of coconut shell biochar.
MATERIALS AND METHODS
Study area and duration: The study was conducted in Benin City, Edo State, Nigeria (approximately 6.34°N, 5.63°E), located in the southern region of the country.
Sample collection: Coconut (Cocos nucifera) shell (about 2 kg) was obtained from Uselu, Benin City. The coconut shells were thoroughly washed with distilled water to remove adhering dirt, and then air-dried for 7 days. The dried shells were broken into pieces, crushed and then sieved into particles of size ≤2 mm. Approximately 500 g of the prepared coconut shell was used for the production of biochar. The experimental work was carried out from the period of February, 2023 to October, 2024.
Preparation of biochar: Slow pyrolysis is performed at various maximum heat treatment temperatures (HTT), 350, 400, 500, 600, 700°C, and maintained for 30 min was used to produce the coconut shell biochar. The resulting biochars were designated CSB350, CSB400, CSB500, CSB600, and CSB700. The biochar samples were then ground before use.
Biochar pH: A mixture of biochar and deionized water (1:10 wt/wt) was prepared, and the slurry was equilibrated for 1 hr before measuring the pH using a precalibrated digital pH meter (Jenway 3020, DUNMOW ESSEX, JENWAY LTD., England).
Carbon yield: The amount of carbon retained in the biochar (carbon yield) was estimated using Eq. 17:
| (1) |
Here, Y(carbon) and Y(mass) represent the carbon yield and mass yield of biochar, respectively, and Cm and Ccb are the carbon content of the original shell and the carbon of the resultant biochar, respectively
Chemical oxidation stability: The KMnO4 method11 was used to assess the easily oxidizable fraction of the studied biochars. Biochar samples were acid pretreated to remove inorganic carbonates following the method described by Fidel et al.12. Typically, the biochar was added to a solution of KMnO4 (33mM, pH 7.2) in a 1:50 w/v biochar: KMnO4 ratio. It was agitated for 60 min, heated at about 60°C for 60 min and thereafter left overnight at 25°C. The solid residue was separated, thoroughly rinsed with distilled water, and dried at 80°C for 4 days. The mass loss from the oxidation treatment was measured gravimetrically, and the dried sample was subsequently analyzed for its elemental composition (C, H, N, O, and S).
An indication of the stability of biochar to oxidative degradation, AE may be obtained from Eq. 2:
| (2) |
Where, BrC and BtC represent the carbon content in the residual and total biochar, respectively, and Br and Bt are the mass after and before treatment5.
Analytical methods: Ultimate analysis was performed using a Varian MACRO Elementar (Germany), with oxygen content determined by difference.
RESULTS AND DISCUSSION
Yield and pH of biochar: The variation of biochar yield with pyrolysis temperature is given in Table 1. The results show that a decrease with an initial increase in HTT, from 67.70% for CSB350 to 53.30% for CSB400, may be attributed to the mass loss as a result of thermal decomposition of cellulose and hemicellulose. Beyond this point, further increases in HTT, a more gradual decrease in the biochar yield, which corresponds to the breakdown of the highly branched lignin component of the biomass during pyrolysis, was observed. The results in Table 1 show that an increase in HTT was associated with a decrease in biochar yield from 53.30 to 35.70%. These results are consistent with those reported in the literature4-13.
Biochar pH increased with heat treatment temperature, from 7.90±0.90 for CSB350 to 10.70±0.77 for CSB700, an increase of 2.8 pH units. Increase in pH of biochar with increase in heat treatment temperature has been attributed to accumulation of inorganic salt (notably carbonates) in the biochar, resulting from a high degree of carbonisation14.
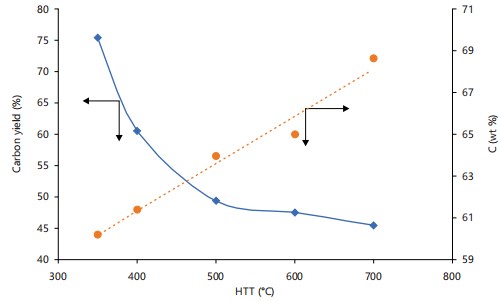
Elemental composition of biochar: The elemental composition of biochar is given in Table 1, and the effect of heat treatment temperature on the carbon yield is shown in Fig. 1.
The range of temperatures used in this work (350 to 700°C) was associated with an increase in carbon (C) content of the biochar from 60.18 to 68.63%. The increase in C content of the biochar as the pyrolysis temperature increases contrasts with the downward trend observed in its yield. This increase in C content could offset the decreasing yield of biochar. Thus, the C content of biochar may provide a better estimate of the effectiveness of carbon sequestration than the yield of biochar. In contrast, the Oxygen (O) content decreased markedly with an increase in heat treatment temperature (HTT). The Hydrogen (H) and Sulphur (S) of the biochar decreased with HTT, while the Nitrogen (N) content increased. The O content decreased markedly with an increase in heat treatment temperature. The variations in the contents of C, H, N, S, and O in biochar at different heat treatment temperatures reflect its stability and resistance to degradation in the environment. The H:C and O:C atomic ratios act as indicators of the biochar’s aromaticity and level of maturation, while the (N+O)/C ratio provides a measure of its polarity15. Table 2 gives the variation of atomic ratios H:C, O:C, and (N+O)/C of the biochar samples with HTT.
| Table 1: | Properties of coconut shell and biochar at different heat treatment temperatures | |||
| Ultimate composition (wt%) | |||||||
| Samples | Biochar yield (%) | Biochar pH | C | H | O | N | S |
| CS | - | - | 54.03±0.00 | 3.22±0.01 | 41.74±0.07 | 0.60±0.04 | 0.43±0.01 |
| CSB350 | 67.70±0.50 | 7.90±0.90 | 60.18±0.02 | 3.03±0.02 | 36.14±0.03 | 0.41±0.02 | 0.22±0.02 |
| CSB400 | 53.30±0.35 | 8.25±1.20 | 61.39±0.02 | 2.99±0.02 | 33.85±0.06 | 0.48±0.00 | 0.24±0.01 |
| CSB500 | 41.67±0.55 | 10.01±0.50 | 63.95±0.03 | 2.53±0.03 | 33.75±1.78 | 0.59±0.00 | 0.40±0.00 |
| CSB600 | 39.44±0.64 | 10.25±0.36 | 65.01±0.00 | 2.43±0.02 | 33.37±0.00 | 0.93±0.01 | 0.083±0.01 |
| CSB700 | 35.76±0.42 | 10.70±0.77 | 68.63±0.01 | 2.30±0.02 | 27.36±0.04 | 0.98±0.03 | 0.037±0.01 |
| H: Hydrogen, O: Oxygen, N: Nitrogen, S: Sulfur and wt%: Weight percentage | |||||||
|
| Table 2: | Atomic mass ratios of coconut shell biochar prepared at different heat treatment temperatures | |||
| Samples | O/C | H/C | (O+N)/C |
| CSB350 | 0.6 | 0.05 | 0.61 |
| CSB400 | 0.54 | 0.05 | 0.55 |
| CSB500 | 0.6 | 0.04 | 0.55 |
| CSB600 | 0.53 | 0.04 | 0.54 |
| CSB700 | 0.4 | 0.03 | 0.41 |
| O/C: Oxygen-to-carbon ratio, H/C: Hydrogen-to-carbon ratio, (O+N)/C: Combined oxygen and nitrogen to carbon ratio, Subscripts (350-700) indicate the pyrolysis temperatures (°C) at which carbonized samples (CSB) were prepared | |||
As shown in the Table 2, the elemental ratios of carbonized samples (CSB) vary with increasing pyrolysis temperature. The O/C and H/C ratios gradually decrease from CSB350 to CSB700, indicating enhanced carbonization and reduced oxygen and hydrogen content at higher temperatures. Similarly, the (O+N)/C ratio declines, suggesting a progressive loss of volatile elements and an increase in aromatic carbon structure as temperature rises.
The H:C and O:C ratios of the coconut shell biochars decreased with an increase in heat treatment temperature, suggesting that the transformation of the lignocellulosic materials into relatively stable C with high aromaticity and saturation in the biochar16,17. The H:C ratio is well below 0.6, meeting the standard set by the proposed European Biochar Certificate (EBC). A decrease in the O:C ratio reflects the loss of oxygen-containing functional groups in the biochar18. Similarly, the decrease in the (N+O)/C ratio suggests reduced polarity and increased hydrophobicity, which can be attributed to the enhanced aromaticity of the biochar19. The results suggest that increase in heat treatment temperature of coconut shell leads to biochar with enhanced stability and recalcitrance and greater potential in carbon sequestration ability.
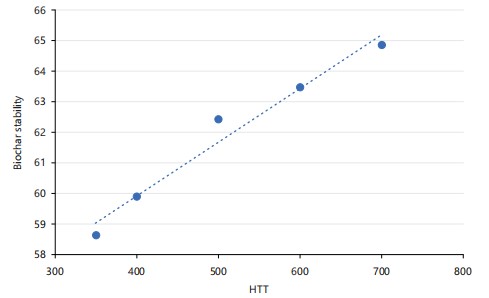
Chemical oxidation stability: Figure 2 illustrates the effect of heat treatment temperature (HTT) on biochar stability. Biochar stability increased progressively with increasing HTT, rising from approximately 58-59 units at ~350°C to about 65 units at ~700°C. The dotted regression line indicates a clear positive linear relationship, suggesting that higher pyrolysis temperatures enhance biochar structural stability, likely due to increased carbonization and formation of more condensed aromatic structures.
It can be seen that oxidation stability increased with heat treatment temperature from 58.63% for CSB350 to 64.84% for CSB700.
|
|
| Table 3: | Elemental analysis of coconut shell biochar after oxidation | |||
| Samples | C (%) | H (%) | O (%) | H/C | O/C | Biochar stability (%) |
| CSB350 | 76.813 | 2.717 | 19.617 | 0.0451479 | 0.325972 | 58.6343 |
| CSB400 | 78.435 | 1.843 | 18.2 | 0.0300212 | 0.296465 | 59.896 |
| CSB500 | 82.32 | 1.283 | 14.7 | 0.0200625 | 0.229867 | 62.4233 |
| CSB600 | 86.57 | 0.445 | 12.487 | 0.0068451 | 0.192078 | 63.4541 |
| CSB700 | 87.987 | 0.963 | 10.583 | 0.0140318 | 0.154204 | 64.8425 |
Elemental composition of coconut shell biochar, together with the H:C and O:C atomic ratios after oxidation, is given in Table 3.
Table 3 shows that increasing the pyrolysis temperature enhanced the carbon content and stability of coconut shell biochar, while hydrogen and oxygen contents, along with H:C and O:C ratios, decreased indicating greater aromaticity and thermal stability at higher temperatures.
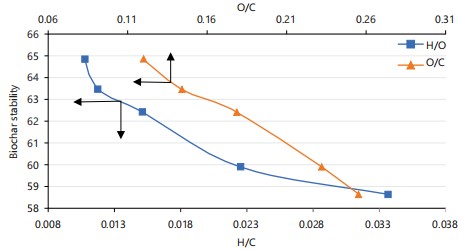
It can be seen that the trend in the decrease of H:C and O:C ratios in the oxidised biochar is similar to the trend in the untreated biochar samples (Table 2). These ratios are used as proxies for degree of aromaticity and maturation15-20. The H/C ratio has also been used as an index and resistance to microbial attack and chemical degradation21; the smaller the H/C ratio, the higher the chemical oxidation stability. The decrease in the O/C ratio is attributed to the loss of surface oxygen functional groups at higher heat treatment temperatures, resulting in the loss of polarity and reduced susceptibility to degradation.
Figure 3 illustrates the relationship between the various indices of biochar: oxidative stability and hydrogen to carbon (H:C) and oxygen to carbon (O:C) atomic ratios. The higher values of biochar stability at low values of H:C and O:C ratios are consistent with the results from previous reports22-24. The increase in biochar stability with increase in heat treatment temperature can be related to the chemical composition and structure of the biomass feedstock and the changes they undergo during pyrolysis. The decomposition of cellulose and hemicellulose component of lignocellulosic materials during pyrolysis occurs more readily and at a relatively low heat treatment temperatures to produce biochar with labile C structures, that can be susceptible to further degradation at higher heat treatment temperatures. Higher heat treatment temperatures result in the loss of labile C structures, increase in aromaticity of the residual C structures and higher resistance to chemical oxidation25-27.
CONCLUSION
This study demonstrated that higher pyrolysis temperatures enhanced the carbon content, aromaticity, and hydrophobicity of coconut shell biochar, as indicated by decreasing H:C and O:C ratios. Carbon stability, measured via KMnO oxidation, ranged between 58.63% and 64.84%, confirming improved oxidative resistance at elevated temperatures. These findings provide a basis for optimizing pyrolysis conditions to produce biochar with tailored stability for long-term environmental applications.
SIGNIFICANCE STATEMENT
This study discovered the influence of pyrolysis temperature on the oxidative stability and chemical properties of coconut shell biochar, which can be beneficial for enhancing its performance in carbon sequestration, soil amendment, and environmental remediation. The research highlights how increasing pyrolysis temperature improves carbon content, aromaticity, and hydrophobicity, all of which are linked to long-term biochar stability. The findings also provide empirical data to support the selection of optimal pyrolysis conditions for producing biochar with desired functional traits. This study will help researchers to uncover the critical areas of biochar stability and functional performance that many researchers were not able to explore. Thus, a new theory on the thermal transformation and stability behavior of biochar may be arrived at.
ACKNOWLEDGMENT
The authors are sincerely grateful to the Department of Chemistry, University of Benin, Benin City, Nigeria for allowing us use some of their facilities.
REFERENCES
- Kizito, S., S. Wu, W.K. Kirui, M. Lei, Q. Lu, H. Bah and R. Dong, 2015. Evaluation of slow pyrolyzed wood and rice husks biochar for adsorption of ammonium nitrogen from piggery manure anaerobic digestate slurry. Sci. Total Environ., 505: 102-112.
- Naisse, C., C. Girardin, R. Lefevre, A. Pozzi, R. Maas, A. Stark and C. Rumpel, 2015. Effect of physical weathering on the carbon sequestration potential of biochars and hydrochars in soil. GCB Bioenergy, 7: 488-496.
- Crombie, K., O. Mašek, S.P. Sohi, P. Brownsort and A. Cross, 2013. The effect of pyrolysis conditions on biochar stability as determined by three methods. GCB Bioenergy, 5: 122-131.
- Chen, D., D. Liu, H. Zhang, Y. Chen and Q. Li, 2015. Bamboo pyrolysis using TG-FTIR and a lab-scale reactor: Analysis of pyrolysis behavior, product properties, and carbon and energy yields. Fuel, 148: 79-86.
- Cross, A. and S.P. Sohi, 2013. A method for screening the relative long-term stability of biochar. GCB Bioenergy, 5: 215-220.
- Wang, T., M. Camps-Arbestain and M. Hedley, 2013. Predicting C aromaticity of biochars based on their elemental composition. Org. Geochem., 62: 1-6.
- Chen, D., X. Yu, C. Song, X. Pang, J. Huang and Y. Li, 2016. Effect of pyrolysis temperature on the chemical oxidation stability of bamboo biochar. Bioresour. Technol., 218: 1303-1306.
- Bakshi, S., C. Banik and D.A. Laird, 2018. Quantification and characterization of chemically-and thermally-labile and recalcitrant biochar fractions. Chemosphere, 194: 247-255.
- Ajien, A., J. Idris, N.M. Sofwan, R. Husen and H. Seli, 2022. Coconut shell and husk biochar: A review of production and activation technology, economic, financial aspect and application. Waste Manage. Res., 41: 37-51.
- Jain, N., A. Bhatia and H. Pathak, 2014. Emission of air pollutants from crop residue burning in India. Aerosol Air Qual. Res., 14: 422-430.
- Tirol-Padre, A. and J.K. Ladha, 2004. Assessing the reliability of permanganate-oxidizable carbon as an index of soil labile carbon. Soil Sci. Soc. Am. J., 68: 969-978.
- Fidel, R.B., D.A. Laird, M.L. Thompson and M. Lawrinenko, 2017. Characterization and quantification of biochar alkalinity. Chemosphere, 167: 367-373.
- Chen, D., J. Zhou and Q. Zhang, 2014. Effects of heating rate on slow pyrolysis behavior, kinetic parameters and products properties of moso bamboo. Bioresour. Technol., 169: 313-319.
- Yuan, J.H., R.K. Xu and H. Zhang, 2011. The forms of alkalis in the biochar produced from crop residues at different temperatures. Bioresour. Technol., 102: 3488-3497.
- Spokas, K.A., 2010. Review of the stability of biochar in soils: Predictability of O:C molar ratios. Carbon Manage., 1: 289-303.
- Han, L., K.S. Ro, Y. Wang, K. Sun, H. Sun, J.A. Libra and B. Xing, 2018. Oxidation resistance of biochars as a function of feedstock and pyrolysis condition. Sci. Total Environ., 616-617: 335-344.
- Harvey, O.R., L.J. Kuo, A.R. Zimmerman, P. Louchouarn, J.E. Amonette and B.E. Herbert, 2012. An index-based approach to assessing recalcitrance and soil carbon sequestration potential of engineered black carbons (biochars). Environ. Sci. Technol., 46: 1415-1421.
- Yang, H., R. Yan, T. Chin, D.T. Liang, H. Chen and C. Zheng, 2004. Thermogravimetric analysis-Fourier transform infrared analysis of palm oil waste pyrolysis. Energy Fuels, 18: 1814-1821.
- Ahmad, M., S.S. Lee, X. Dou, D. Mohan, J.K. Sung, J.E. Yang and Y.S. Ok, 2012. Effects of pyrolysis temperature on soybean stover- and peanut shell-derived biochar properties and TCE adsorption in water. Bioresour. Technol., 118: 536-544.
- Wiedemeier, D.B., S. Abiven, W.C. Hockaday, M. Keiluweit and M. Kleber et al., 2015. Aromaticity and degree of aromatic condensation of char. Org. Geochem., 78: 135-143.
- Kuhlbusch, T.A.J. and P.J. Crutzen, 1995. Toward a global estimate of black carbon in residues of vegetation fires representing a sink of atmospheric CO2 and a source of O2. Global Biogeochem. Cycles, 9: 491-501.
- Malghani, S., G. Gleixner and S.E. Trumbore, 2013. Chars produced by slow pyrolysis and hydrothermal carbonization vary in carbon sequestration potential and greenhouse gases emissions. Soil Biol. Biochem., 62: 137-146.
- Wei, L., Y. Huang, L. Huang, Y. Li and Q. Huang et al., 2020. The ratio of H/C is a useful parameter to predict adsorption of the herbicide metolachlor to biochars. Environ. Res., 184. https://doi.org/10.1016/j.envres.2020.109324
- Leng, L., H. Huang, H. Li, J. Li and W. Zhou, 2019. Biochar stability assessment methods: A review. Sci. Total Environ., 647: 210-222.
- Kuryntseva, P., K. Karamova, P. Galitskaya, S. Selivanovskaya and G. Evtugyn, 2023. Biochar functions in soil depending on feedstock and pyrolyzation properties with particular emphasis on biological properties. Agriculture, 13.
- Petersen, H.I., L. Lassen, A. Rudra, L.X. Nguyen, P.T.M. Do and H. Sanei, 2023. Carbon stability and morphotype composition of biochars from feedstocks in the Mekong Delta, Vietnam. Int. J. Coal Geol., 271.
- Almutairi, A.A., M. Ahmad, M.I. Rafique and M.I. Al-Wabel, 2023. Variations in composition and stability of biochars derived from different feedstock types at varying pyrolysis temperature. J. Saudi Soc. Agric. Sci., 22: 25-34.
How to Cite this paper?
APA-7 Style
Ejimadu,
C.M., Oku,
J.M., Okieimen,
F.E. (2026). Chemical Stability of Coconut Shell Biochar Under Oxidative Conditions. Singapore Journal of Scientific Research, 16(1), 1-7. https://doi.org/10.3923/sjsr.2026.01.07
ACS Style
Ejimadu,
C.M.; Oku,
J.M.; Okieimen,
F.E. Chemical Stability of Coconut Shell Biochar Under Oxidative Conditions. Singapore J. Sci. Res 2026, 16, 1-7. https://doi.org/10.3923/sjsr.2026.01.07
AMA Style
Ejimadu
CM, Oku
JM, Okieimen
FE. Chemical Stability of Coconut Shell Biochar Under Oxidative Conditions. Singapore Journal of Scientific Research. 2026; 16(1): 1-7. https://doi.org/10.3923/sjsr.2026.01.07
Chicago/Turabian Style
Ejimadu, Chima, Maximus, James Majebi Oku, and Felix Ebhodaghe Okieimen.
2026. "Chemical Stability of Coconut Shell Biochar Under Oxidative Conditions" Singapore Journal of Scientific Research 16, no. 1: 1-7. https://doi.org/10.3923/sjsr.2026.01.07

This work is licensed under a Creative Commons Attribution 4.0 International License.


